Aren’t phosphate, phosphorus, and phosphorous just variants of the same thing? Absolutely not. While they all share roots in their meanings, these three terms should be used correctly. So, buckle up as I break it all down for you.
Phosphate vs. Phosphorus vs. Phosphorous
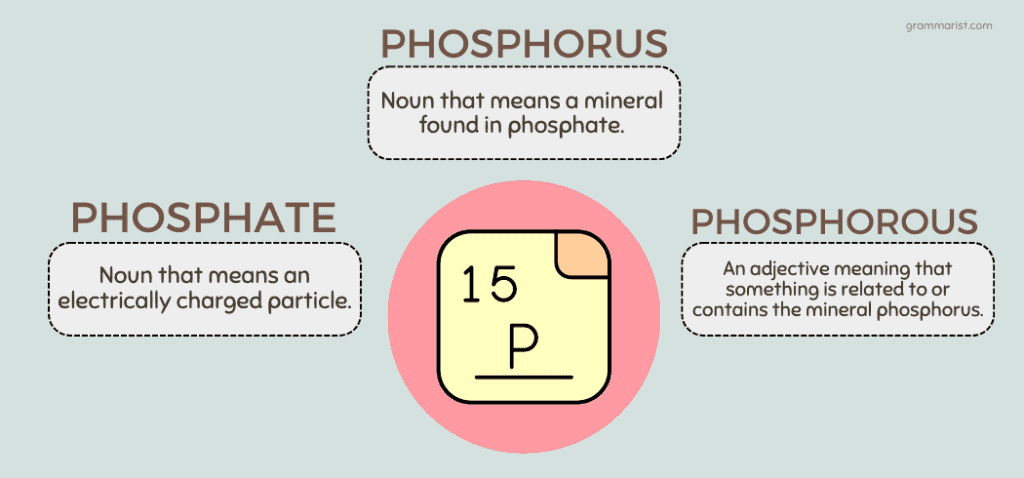
Could three words be similar? You might think they’re all variant spellings of the same term, but you’d be wrong. Each of these has its own definition. Instead of explaining it away, I’ll put it in three simple points for you.
- Phosphate: Noun that means an electrically charged particle.
- Phosphorus: Also a noun that means a mineral found in phosphate.
- Phosphorous: An adjective meaning that something is related to or contains the mineral phosphorus.
Here’s a zinger to get your brain tingling that contains all three words. “The phosphate contains phosphorus, which makes the soil phosphorous.” Make sense?
What Does Phosphate Mean?
To put it in simple terms, it’s a noun, and it’s a particle that’s been electrically charged. You can find phosphate ions in our bones and teeth because the mineral found within them (phosphorus) mixes with other minerals like calcium (becoming calcium phosphate) and to make our skeleton strong.
It’s not to be confused with phosphoric acid, a chemical compound found in detergents and fertilizers.
What Does Phosphorus Mean?
As I mentioned above, phosphorus is the mineral that’s inside phosphates. Like, if you went to the doctor and had some bloodwork done and the doc told you that you had a deficiency in phosphorus, he’d probably prescribe some supplements with phosphorus in them.
There are also different kinds of phosphorus, like white phosphorus and red phosphorus. The chemical element symbol of phosphorus is P.
What Does Phosphorous Mean?
I know what you’re thinking; it’s the UK spelling of phosphorus. But you’d be wrong! Even though they’re spelled exactly the same, with the exception of one U, they’re actually homophones.
Phosphorus is a noun, as I said earlier, which is a mineral. But phosphorous, with the extra U at the end, is an adjective we use to describe something with phosphorus.
Difference Between Phosphate and Phosphorus
One is a particle (phosphate), and the other is a mineral (phosphorus) that exists within the particle.
Which Is Correct Phosphorus or Phosphorous?
When it comes to phosphorus and phosphorous, both are correct if you’re asking which one is an actual word. But they have their own definitions and are not just alternative spellings of the same word. Here, I’ll put it simply:
- Phosphorus: Mineral
- Phosphorous: The effect of the mineral
Are Phosphate and Phosphorous the Same?
No, they’re definitely not. I know it’s confusing because they look so similar. Phosphate is the noun you’d use to describe the particle in science. Phosphorous is the adjective you’d use when describing how something is high in the mineral phosphorus.
Phosphor vs. Phosphorus
We’ve already established that phosphorus is the simple mineral found in the particle phosphate, but phosphor is something else altogether. It’s a noun and means a substance that is luminescent when exposed to radiation. They use it in fluorescent lamps and stuff.
Phosphorus Spelling

When spelling the noun that means the phosphorus mineral, you spell it as p-h-o-s-p-h-o-r-u-s.
Using Phosphate in a Sentence
- Most phosphates were left behind by decaying marine microorganisms.
- Aluminum hydroxide and phosphates are better alternatives to some chemicals used in our food.
Using Phosphorus in a Sentence
- The soil is rich with phosphorus, so it’ll be great for growing our veggies in the spring.
- We need phosphorus to help our teeth and bones grow strong.
- The dentist said I lack phosphorus in my system, which is why my bones break so easily.
Using Phosphorous in a Sentence
- That soil is almost too phosphorous; you need to treat it with other minerals.
- Those compounds are too phosphorous for this experiment.
Final Words
There you go. I hope I explained the key differences between the terms phosphate, phosphorus, and phosphorous. I know it can get confusing, so just refer back to this guide whenever you’re unsure.

Comments are closed.